In the field of drug development, the journey from lab discoveries to new treatments reaching the market is long and complex. Clinical trials play a crucial role in this journey: they bridge the gap between theory and practice.
However, traditional methods of conducting clinical trials can also be complicated. Problems with inefficient data collection and slow processing times can plague trials.
New, innovative clinical trial technology is helping to revolutionize the research landscape. And a big factor in this shift was COVID-19. In order to deliver safe and effective vaccines, companies suddenly needed to condense years of clinical trials into just a few months. The concept of automation through technology was immediately appealing.
Using technology, nearly every element of COVID-19 clinical trial design, data collection, analysis and approval was shortened. Technology helped ensure the swift rollout of safe and effective vaccines. It also demonstrated that trials can be run much more efficiently and faster than the industry previously imagined – without compromising quality
Before we look at these improvements in more depth, let’s take a look at the challenges of traditional clinical trial processes.

Challenges in traditional clinical trial processes
Traditionally, clinical trials are long and time-consuming. According to Cancer Research UK, ‘There is no typical length of time it takes for a drug to be tested and approved. It might take 10 to 15 years or more to complete all 3 phases of clinical trials before the licensing stage.’ And, according to the US Congressional Budget Office, only ‘about 12 percent of drugs entering clinical trials are ultimately approved for introduction by the FDA’.
The pharmaceutical industry is typically risk-adverse. With new regulations coming out all the time, researchers often find themselves caught up in paperwork, delaying the start of trials and wasting valuable time. Typically, researchers have to navigate long internal procurement processes, and regulatory time pressures. In this environment, the idea of implementing new processes or innovations can seem risky. It can feel that the ‘right time’ to re-assess current processes just never comes.
But there are some problems with traditionally run clinical trials. Here’s just a few:
➡️ Time-consuming manual processes
While paper-based studies are becoming a thing of the past, the use of manual solutions such as Excel spreadsheets and Word documents is still common practice in designing and building trials.
While these solutions might feel comfortable (things have been done that way for a long time, so why change them?) these processes are risky too. The likelihood of human error or misinterpretation of data is high. It can also be difficult to find information that’s stored in disparate systems, each with their own ownership, access rights and permissions.
How can you know you’re using the right case report form template, for example?
And how can an Excel spreadsheet help you enforce CDISC standards?
➡️ Ineffective change management
Change management is problematic when using these formats: it can be difficult for data management teams to see the downstream impact of making changes to content. Or to identify if they are working on the most up-to-date, correct version of a particular document.
➡️ No real-time monitoring or insights
Real-time monitoring, or visibility of collected data, is almost impossible when using manual processes. For example, you cannot check early in the process that all the necessary data is being collected, in the format required for submission to the regulators. It isn’t uncommon for organizations to find out, after the trial is finished, that their collected data isn’t up to scratch.
Clinical trial technology can be used to automate traditionally manual processes, and to help produce readily submittable data. It can enable early data insights, and ensure CDISC compliance. A large part of what makes this possible is metadata standardization.
Metadata standardization is key
There has been huge progress in recent years towards standardizing data for clinical trial submissions, including the development of the Clinical Data Interchange Standards Consortium (CDISC) standards. These are now used by several regulatory bodies, including the Food and Drug Administration (FDA) and the Japanese Pharmaceuticals and Medical Devices Agency (PMDA) for submission packages.
CDISC standards provide guidance and structure for the end-to-end collection and submission of clinical trial data. The creation of these standards was influenced by some persistent challenges faced by those companies still applying manual processes – like the ones we mentioned above.
Lack of metadata standardization in clinical trial design leads to many of these problems. Metadata is often not stored centrally or controlled effectively. And because it’s not standardized it can’t be reused, which means that each trial needs to be set up manually from individual specifications. Not only does this introduce the risk of errors and inconsistencies, but it also extends timelines due to the manual work needed.
Using the standards as a guide, organizations can create standardized, approved metadata such as forms, mappings, and terminologies. This content can then be used and reused for faster clinical study build.
All-in-one cloud-based platforms can be used to simplify end-to-end metadata management, enabling cross-collaboration between stakeholders. Ultimately, by utilizing clinical trial technology, organizations can benefit from significant time and cost savings, resulting in the faster delivery of new therapies to patients.
The role of clinical trial technology in streamlining research processes
With the right platform, studies can be built faster, and data analysis can be performed more quickly. As a result, researchers can identify patterns, generate insights, and make data-driven decisions about the safety and efficacy of new treatments.
One way to manage your metadata is by using a clinical metadata repository (CMDR). A CMDR is a centralized library where you can store, manage and easily search for all your study metadata.
Metadata can not only be stored in the repository, it can be updated, developed and approved there. This provides easy access to approved metadata that you can reuse again and again. This means less time spent creating and approving metadata with every study.
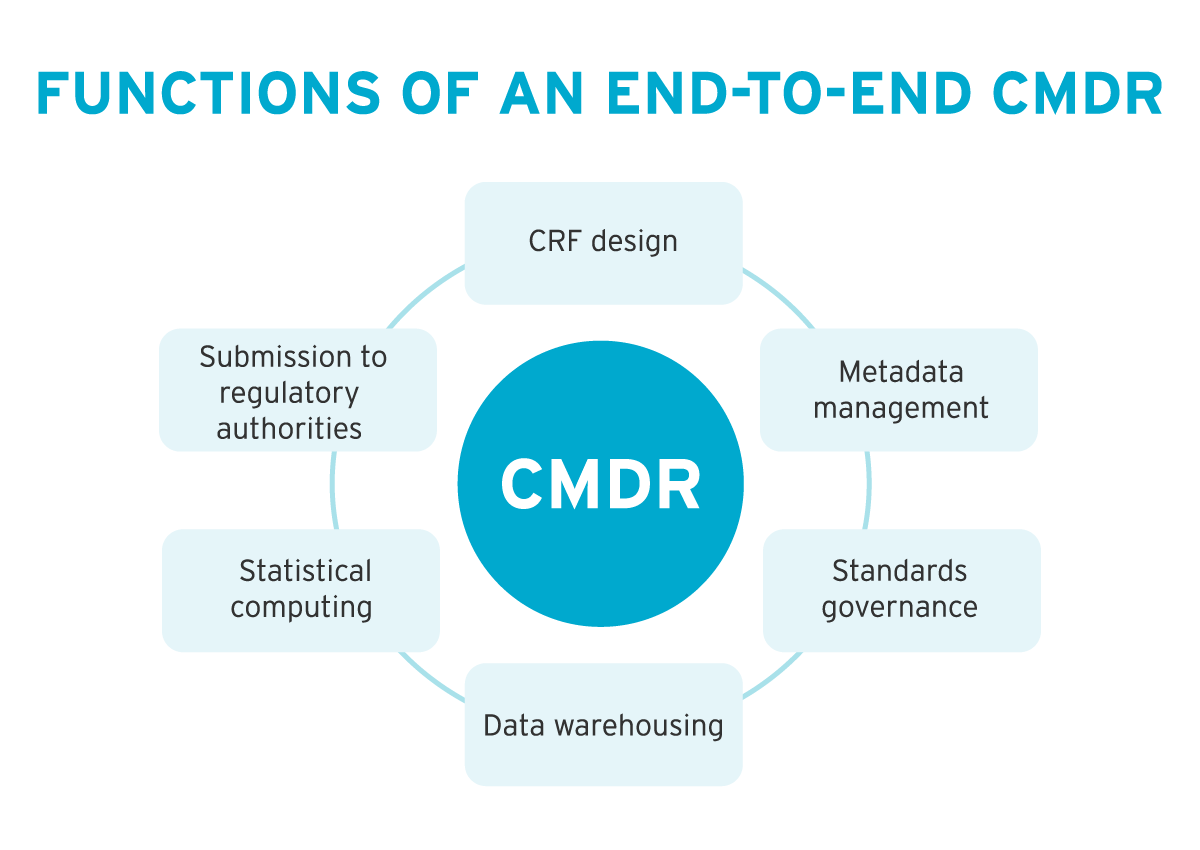
Functions of an end-to-end CMDR:
- CRF design
- Metadata management
- Standards governance
- Data warehousing
- Statistical computing
- Submission to regulatory authorities
➡️ Real-time data
CMDRs enable real-time data access and analysis. By embedding end-to-end standards throughout the clinical trial design, data collection and analysis stages, submission-ready datasets can be generated as soon as data starts rolling in.
Researchers can monitor data trends, identify outliers, and make timely decisions based on the insights gained. This not only speeds up the overall trial process but also allows for early detection of adverse events or unexpected outcomes, ensuring participant safety.
➡️ Seamless collaboration
A central, single source for metadata in the form of a CMDR also encourages a collaborative approach, rather than a silo mentality. Stakeholders can access one system and view the most current versions of the relevant metadata and know that it is controlled and standardised.
Using a CMDR, reviews and approvals take place centrally, rather than through multiple spreadsheets and email chains. Therefore, the full history of a standard can be traced throughout the system, with every sign-off and change documented.
Electronic data capture (EDC) systems
EDC systems can be used hand-in-hand with CMDRs. EDC systems allow researchers to collect, store, and manage clinical trial data electronically, eliminating the need for manual data entry and reducing the risk of errors. This not only saves time but also improves data accuracy and integrity.
With the right CMDR in place, EDC automation allows teams to preview exactly how their forms will look for their chosen EDC system. By standardizing data and allowing team members to collaborate in one system, human error and miscommunication are reduced. And, with clear protocols for change control, impacts can be studied before a change is made, which reduces complications downstream too.
The best part is once your team has approved your study metadata, the automation software automatically builds the study and converts your data as it is collected.

Case studies of successful implementation of clinical trial technology
Numerous case studies showcase the successful implementation of clinical trial technology in various research settings. Here are some examples:
Case study 1
In 2019, we collaborated with a UK-based consortium interested in observing early safety data. They used ryze, our cloud-based CMDR, extensively in several COVID-19 vaccine trials, to reduce the lag time for SDTM conversions. Raw data was quickly converted to SDTM formats and visualized, allowing early assessments of vaccine safety and efficacy.
COVID-19 taught the industry a lot and brought expectations to a new level. It’s hard to imagine that pharmaceutical companies will return to the manual and lengthy processes of the past.
Case study 2
In another case, a large Boston-based biopharmaceutical organization chose the ryze platform as their CMDR, with the aim of standardizing all data collection metadata including EDC specific content.
They have used ryze for all their EDC designs since 2020. One team member built three unique studies in three months, from protocol to go-live.
Following their successful adoption of CMDR technology, they have since expanded their use of ryze to incorporate dataset metadata in 2022, from standardized dataset mapping specifications to study specific datasets.
.png?width=1500&height=804&name=3-studies---3-months-graphic%20(1).png)
The transformative impact of clinical trial technology
Clinical trial technology, including CMDRs and EDCs, has proven to be a game-changer in the field of clinical research. The streamlining of traditional processes has enabled researchers to implement efficiencies and make the journey from lab discoveries to the availability of life-saving treatments easier and faster.
With advancements in clinical trial technology, researchers can now streamline their clinical trial processes, leading to more accurate data collection and faster submission. From metadata repositories to electronic data capture systems, innovative technologies are paving the way for more efficient clinical trials. For more information, check out our blog to how clinical trial software can help to optimize clinical trials.
Want to read more about CMDRs? Why not download our free whitepaper: 'The importance of a clinical metadata repository in clinical trials'?
![]()
About the author

Gilbert Hunter
Customer Success Manager | Formedix
Gilbert joined Formedix nearly ten years ago as a technical writer. The system knowledge he gained from content development, together with his existing customer service skills, marked him out for transition to the Professional Services (PS) team.
Gilbert has worked with the PS team for over four years, providing both CDISC-based training and software training, as well as support and consultancy services to Pharmaceutical, Biotechnology and Clinical Research Organizations. He helps organizations build studies faster and to a higher quality by making their clinical trial design and regulatory submissions far more efficient.
Today, as Customer Success Manager, Gilbert’s focus is to ensure customers maximize the benefits they can achieve from ryze by overcoming their challenges and achieving their goals.









